Similar to land plants, marine algae are also more likely to assimilate the
lighter carbon isotope 12C over the heavier 13C, and the
carbon isotopic composition of their biomolecules, δ13Cphyto,
is therefore ~10-25‰ lower than the δ13C of dissolved
CO2 in surface seawater. Assuming most of the CO2 arrives
at the site of photosynthesis by diffusion through the algal cell membranes,
the degree to which algae discriminate against 13C, commonly expressed
as εp, depends on the concentration of their aqueous carbon source;
the more carbon is available, the more selective the algae can be and this will
lower the δ13Cphyto (see Figure). This basic theory
has been confirmed in laboratory and field experiments, but the same experiments
have also identified a number of additional factors that can affect
δ13Cphyto, including growth rate, cell size,
irradiance, and the actual carbon source, i.e. carbon dioxide (CO2)
or bicarbonate ion (HCO3-). For instance, the faster algae
grow, the less selective they are in their carbon isotope uptake, so that
δ13Cphyto increases. There have been several attempts
to approximate the degree to which growth rate and other physiological processes
modify the original [CO2] signal. In their simplest form, physiological
influences have been summarized by an empirical ‘b’ factor, which relates to
the carbon-specific and enzymatic carbon isotope fractionation as follows:
εp = εf - b/[CO2]
where εp is the carbon isotope fractionation between aqueous CO2
and δ13Cphyto (εp ≈ δ13CCO2– δ13Cphyto), and εf
is the carbon isotope fractionation associated with enzymatic carbon fixation,
which is typically assumed to be constant at 25‰. Using this framework,
paleo-CO2 can be estimated when δ13Cphyto
data can be paired with independent estimates of temperature, cell size, growth
rate and δ13CCO2. It should also be noted that one
of the major advantages of the proxy is that the 13C/12C
ratio of biomarkers tends to be preserved well in ocean sediments and thereby
alleviates one of the concerns associated with many paleoproxies, i.e. the
potential modification of the original proxy signal through diagenetic processes.
For additional information, see also General background and some commonalities
of paleo-CO2 proxies.
The first paleoreconstructions using the εp proxy analyzed bulk organic
matter (i.e. all organic carbon in a sample). However, the mixture of different
kinds of algae in bulk organic matter quickly led researchers to focus on long-chain
alkenones (Jasper and Hayes, 1990) which are biomolecules that are produced by a
specific group of marine algae, the haptophytes. Although alkenones are found
throughout the Cenozoic and even the Cretaceous (cf. Brassell, 2014) the current
alkenone-based CO2 reconstructions are restricted to the past ~40
million years, when alkenones were more abundant in deep-sea sediments
(Pagani et al., 2005b; Zhang et al., 2013).
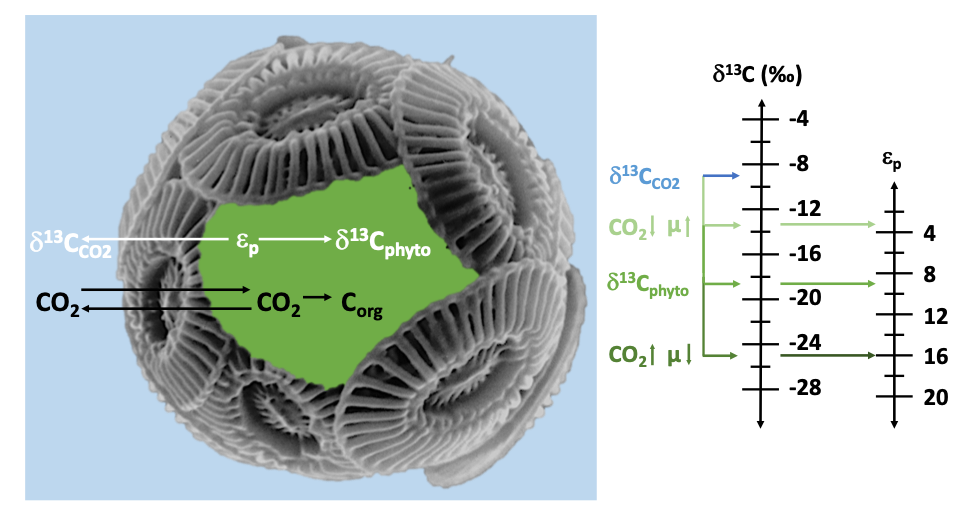 The basic theory of the phytoplankton δ13C proxy
relates the δ13C of algae biomarkers to the δ13C
of the aqueous carbon source for photosynthesis. This relationship is summarized
in εp, the carbon isotope fractionation associated with photosynthetic
carbon fixation. εp increases when aqueous carbon supply (i.e. CO2)
is high, but also when algae growth rates (μ) are slow.
The basic theory of the phytoplankton δ13C proxy
relates the δ13C of algae biomarkers to the δ13C
of the aqueous carbon source for photosynthesis. This relationship is summarized
in εp, the carbon isotope fractionation associated with photosynthetic
carbon fixation. εp increases when aqueous carbon supply (i.e. CO2)
is high, but also when algae growth rates (μ) are slow. Despite generally convincing CO2 estimates during geologic periods of globally warmer climates, comparisons between ice-core and alkenone-derived CO2 estimates over the past 800,000 years suggest that the latter perform poorly, and have led to questions about the accuracy and validity of the diffusive uptake of carbon by algae. The presence of active carbon concentrating mechanisms has been invoked by some investigators (Badger et al., 2019; Stoll et al., 2019) to explain the low sensitivity of alkenone-δ13C to the low CO2 levels of the past ~20 million years. Others (Zhang et al., 2019), however, have suggested that study sites with low modern b values are inherently biased by lower sensitivity of εp37:2 to changing CO2, and that with proper site selection and constraints on the physiological b parameter, it will be possible to reconstruct relatively small atmospheric CO2 changes of the late Pleistocene from alkenones.
The systematics of the alkenone δ13C proxy response to past CO2 levels are an area of active research today and ongoing validation efforts center on evaluating the relative significance of vital effects associated with haptophyte species assemblages, cell size, growth rate, carbon concentrating mechanisms (Badger et al., 2019; Bolton et al., 2016; Bolton and Stoll, 2013; Stoll et al., 2019; Zhang et al., 2019), and the fundamental systematics of carbon isotope fractionation in algae (Boller et al., 2011; Wilkes and Pearson, 2019). Although less research has been done to identify and evaluate controls other than aqueous CO2 on other algae biomarkers, paleo-CO2 has been estimated from such biomarkers across the entire Phanerozoic (Freeman and Hayes, 1992; Naafs et al., 2016; Pancost et al., 2013; Sinnighe Damsté et al., 2008; Witkowski et al., 2018). Based on the information currently available for the alkenone proxy, other biomarker reconstructions will have to undergo a similar level of scrutiny to be evaluated in detail. Depending on the outcome of ongoing studies, re-calculation of paleo-CO2 from previously published data may change absolute CO2 values from this proxy. However, the major trend in Cenozoic CO2 estimates from this proxy, i.e. higher CO2 prior to 20 million years ago and lower CO2 levels thereafter, is unlikely to change and agrees well with other proxy estimates for CO2 over this time interval.
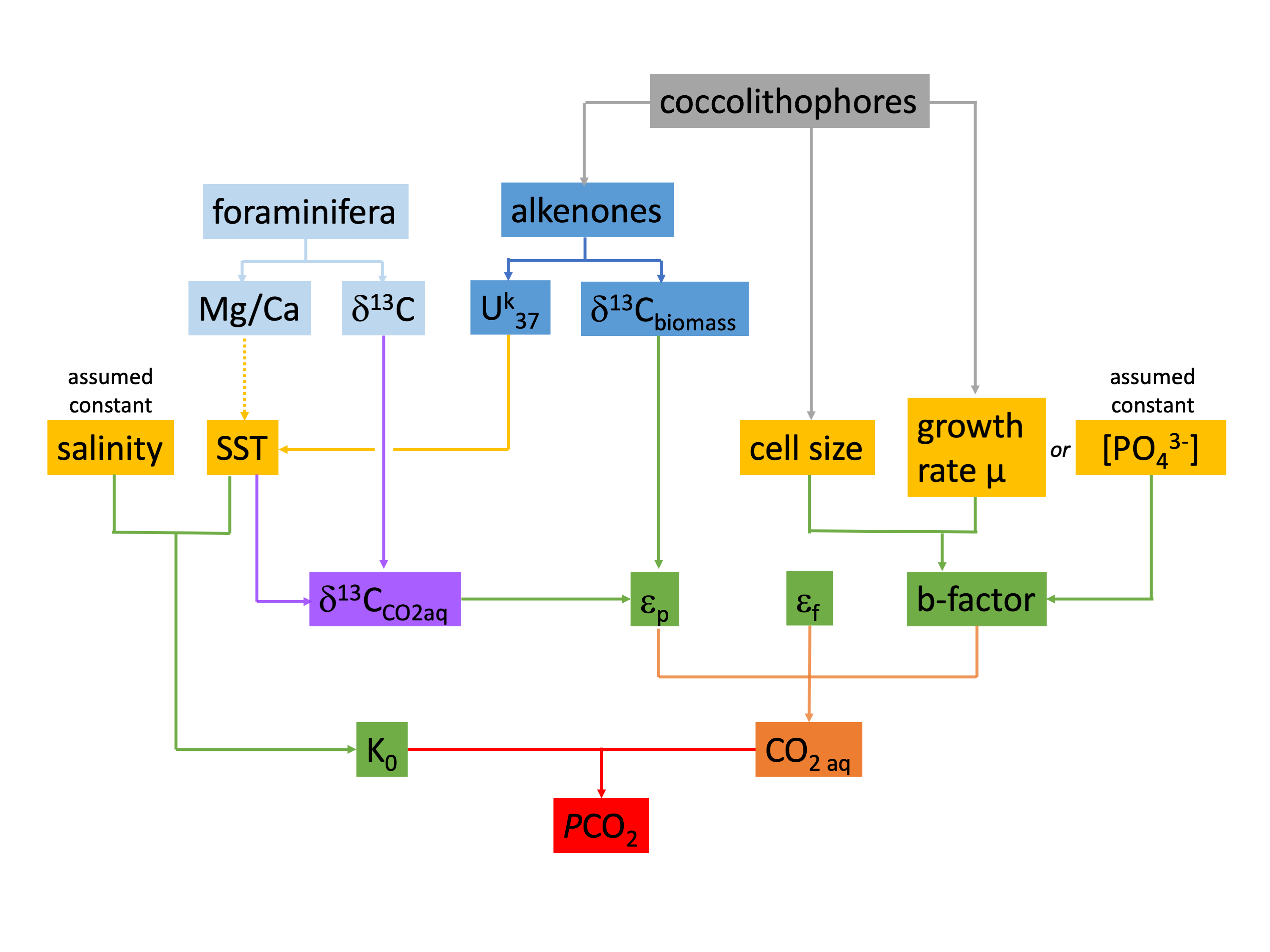 Alkenone flow chart
Alkenone flow chart References cited
Badger, M.P.S., Chalk, T.B., Foster, G.L., Bown, P.R., Gibbs, S.J., Sexton, P.F., Schmidt, D.N., Pälike, H., Mackensen, A. and Pancost, R.D. (2019) Insensitivity of alkenone carbon isotopes to atmospheric CO2 at low to moderate CO2 levels. Clim. Past 15, 539-554.
Boller, A.J., Thomas, P.J., Cavanaugh, C.M. and Scott, K.M. (2011) Low stable carbon isotope fractionation by coccolithophore RubisCO. Geochimica et Cosmochima Acta 75, 7200-7207.
Bolton, C.T., Hernandez-Sanchez, M.T., Fuertes, M.-A., Gonzalez-Lemos, S., Abrevaya, L., Mendez-Vicente, A., Flores, J.-A., Probert, I., Giosan, L., Johnson, J. and Stoll, H.M. (2016) Decrease in coccolithophore calcification and CO2 since the middle Miocene. Nat. Commun. 7.
Bolton, C.T. and Stoll, H.M. (2013) Late Miocene threshold response of marine algae to carbon dioxide limitation. Nature 500, 558-562.
Brassell, S.C. (2014) Climatic influences on the Paleogene evolution of alkenones. Paleoceanography 29, 2013PA002576.
Freeman, K.H. and Hayes, J.M. (1992) Fractionation of carbon isotopes by phytoplankton and estimates of ancient CO2 levels. Glob. Biogeochem. Cycle 6, 185-198.
Jasper, J.P. and Hayes, J.M. (1990) A carbon isotope record of CO2 levels during the late Quaternary. Nature 347, 462-464.
Naafs, B.D.A., Castro, J.M., De Gea, G.A., Quijano, M.L., Schmidt, D.N. and Pancost, R.D. (2016) Gradual and sustained carbon dioxide release during Aptian Oceanic Anoxic Event 1a. Nature Geosci 9, 135-139.
Pagani, M., Huber, M., Liu, Z., Bohaty, S.M., Henderiks, J., Sijp, W., Krishnan, S. and DeConto, R.M. (2011) The Role of Carbon Dioxide During the Onset of Antarctic Glaciation. Science 334, 1261-1264.
Pagani, M., Lemarchand, D., Spivack, A. and Gaillardet, J. (2005a) A critical evaluation of the boron isotope-pH proxy: The accuracy of ancient ocean pH estimates. Geochimica et Cosmochimica Acta 69, 953-961.
Pagani, M., Liu, Z., LaRiviere, J. and Ravelo, A.C. (2010) High Earth-system climate sensitivity determined from Pliocene carbon dioxide concentrations. Nature Geosci 3, 27-30.
Pagani, M., Zachos, J.C., Freeman, K.H., Tipple, B. and Bohaty, S. (2005b) Marked Decline in Atmospheric Carbon Dioxide Concentrations During the Paleogene. Science 309, 600-603.
Pancost, R.D., Freeman, K.H., Herrmann, A.D., Patzkowsky, M.E., Ainsaar, L. and Martma , T. (2013) Reconstructing Late Ordovician carbon cycle variations. Geochimica et Cosmochimica Acta 105, 433-454.
Sinnighe Damsté, J.S., Kuypers, M.M.M., Pancost, R.D. and Schouten, S. (2008) The carbon isotopic response of algae, (cyano)bacteria, archaea and higher plants to the late Cenomanian perturbation of the global carbon cycle: Insights from biomarkers in black shales from the Cape Verde Basin (DSDP Site 367). Organic Geochemistry 39, 1703-1718.
Stoll, H.M., Guitian, J., Hernandez-Almeida, I., Mejia, L.M., Phelps, S., Polissar, P., Rosenthal, Y., Zhang, H. and Ziveri, P. (2019) Upregulation of phytoplankton carbon concentrating mechanisms during low CO2 glacial periods and implications for the phytoplankton pCO2 proxy. Quaternary Science Reviews 208, 1-20.
Super, J.R., Thomas, E., Pagani, M., Huber, M., O’Brien, C.L. and Hull, P.M. (2018) North Atlantic temperature and pCO2 coupling in the early-middle Miocene. Geology 46, 519-522.
Wilkes, E.B. and Pearson, A. (2019) A general model for carbon isotopes in red-lineage phytoplankton: Interplay between unidirectional processes and fractionation by RubisCO. Geochimica et Cosmochimica Acta 265, 163-181.
Witkowski, C.R., Weijers, J.W.H., Blais, B., Schouten, S. and Sinninghe Damsté, J.S. (2018) Molecular fossils from phytoplankton reveal secular pCO2 trend over the Phanerozoic. Science Advances 4.
Zhang, Y.G., Pagani, M., Liu, Z., Bohaty, S.M. and DeConto, R. (2013) A 40-million-year history of atmospheric CO2. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences 371.
Zhang, Y.G., Pearson, A., Benthien, A., Dong, L., Huybers, P., Liu, X. and Pagani, M. (2019) Refining the alkenone-pCO2 method I: Lessons from the Quaternary glacial cycles. Geochimica et Cosmochimica Acta 260, 177-191.